Interpretation of 10 Clinical Trials: Evidence-Based Medical Evidence for the Application of S-Equol in Menopause
Menopause is a natural stage that every woman experiences. Issues such as hot flushes, night sweats, sleep disturbances, mood swings, and compromised bone health have severely impacted the quality of life for many women. While conventional hormone replacement therapy (HRT) is effective, its potential risks (e.g., stimulation to the breast and uterus) deter some women from pursuing it.
Against this backdrop, S-Equol—a gut metabolite of soy isoflavones—has captured scientists’ attention as a mild and promising alternative, owing to its highly selective activation of estrogen receptor beta. Grounded in this scientific principle, we have leveraged advanced biosynthesis technology to achieve green, large-scale production of S-Equol with high purity (≥99%) and high assay (≥99%), committed to supplying high-quality core raw materials for menopausal health.
From 2009 to 2025, more than 25 clinical studies on S-Equol have been published or registered worldwide, covering research regions including China, the United States, Japan, Australia, the United Kingdom, and Singapore. These studies have systematically evaluated the application value of S-Equol across multiple dimensions of health in both women and men. This paper summarizes the findings of 10 clinical trials focusing on S-Equol for menopausal health in women, spanning from 2011 to 2022 and conducted in Japan, the United States, and Australia. It aims to interpret its real value in menopausal health management from an evidence-based medical perspective.
I. Core Effects: Alleviating Hot Flushes, Night Sweats and Sleep Disturbances
Hot flushes and night sweats are the most common and distressing typical manifestations of vasomotor symptoms (VMS) during menopause. Multiple studies have confirmed the potential of S-Equol in this regard.
1. Landmark Study: Validation in Japanese Women
A multicenter, double-blind, placebo-controlled trial published in the Journal of Women’s Health in 2012 enrolled 160 postmenopausal Japanese women who were non-equol producers. The results showed that daily administration of 10 mg of natural S-Equol for 12 weeks led to a significantly greater reduction in hot flush frequency compared with the placebo group (-58.7% vs. -34.5%), while the severity of neck and shoulder muscle stiffness was also markedly alleviated. This was the first landmark trial confirming that 10 mg/day of S-Equol effectively relieves menopausal symptoms, providing key clinical evidence for the oral dosage of 10 mg/day.
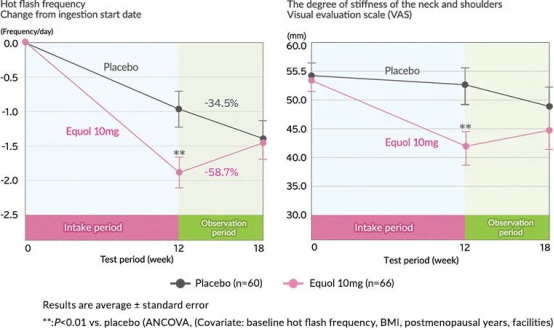
Figure: Effects of S-Equol on hot flushes (left) and neck/shoulder stiffness (right) in menopausal women.
2. Dosage Exploration: Superior Efficacy in Subgroup Analysis
In the same year, an 8-week randomized, double-blind, active-controlled trial in the United States compared the efficacy of different dosages of S-Equol (10 mg, 20 mg, 40 mg/day) with soy isoflavones. The study found that for women with moderate to severe symptoms experiencing more than 8 hot flushes per day, S-Equol at a daily dosage of ≥20 mg was superior to soy isoflavones in reducing hot flush frequency. Additionally, the 10 mg and 20 mg dosage groups showed better outcomes in improving muscle and joint pain.
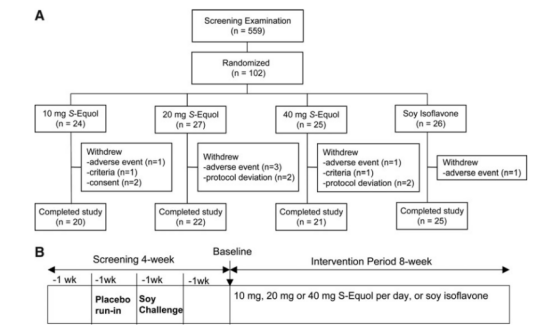
Figure: Subject flow chart (A) and trial design (B).
3. Large-Scale Observational Study: Broad Symptom Improvement
An open-label trial involving 247 menopausal women in 2019 demonstrated that daily supplementation with 10 mg of S-Equol resulted in at least one improved menopausal symptom in 90% of women after 12 weeks. Among these responders, 82%, 71%, and 40% reported improvements in hot flushes, night sweats, and sleep disturbances, respectively, with over 90% describing their symptoms as “reduced”.
4. Comprehensive Improvement: Dual Benefits for Metabolism and Symptoms
A Japanese randomized controlled trial in 2021 found that after 3 months of supplementation with S-Equol (10 mg/day), the improvement rate of menopausal symptoms in the treatment group was significantly higher than that in the control group (81% vs. 53%). The study also proposed a novel hypothesis: equol supplementation may act synergistically with the body’s endogenous equol-producing capacity, thereby improving skin aging and visceral fat in certain populations.
II. Extended Applications: Focus on Bone, Cardiovascular Health and Mood
The effects of S-Equol extend beyond relieving hot flushes; it also exhibits positive protective effects on women’s long-term health, such as bone and cardiovascular health.
1. Bone Health: Inhibiting Bone Resorption and Maintaining Bone Mineral Density
A 12-month Japanese randomized controlled trial in 2011, conducted on postmenopausal non-equol-producing women, found that daily supplementation with 10 mg of S-Equol significantly reduced urinary deoxypyridinoline (a bone resorption marker) by 23.94%, compared with only a 2.87% reduction in the placebo group. This indicates that S-Equol can markedly inhibit bone resorption and help maintain bone health, with no significant impact on serum sex hormones, highlighting its unique safety advantage as a non-hormonal active ingredient.
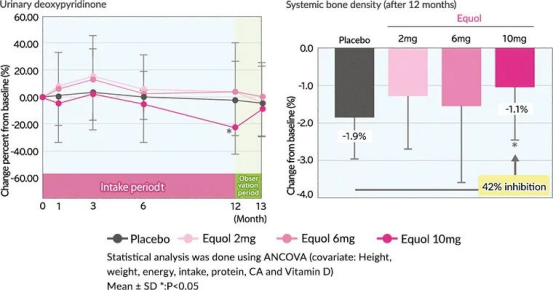
Figure: Effects of different dosages of S-Equol on bone metabolic markers and bone mineral density. At month 12, the decline in total body bone mineral density was significantly inhibited in the 10 mg equol group compared with the placebo group.
2. Cardiovascular Parameters: Improving Arterial Stiffness
A 12-month prospective observational study in 2018 further confirmed that daily supplementation with 10 mg of S-Equol significantly reduced arterial stiffness in middle-aged women, with more pronounced effects in high-risk populations (e.g., patients with hypertriglyceridemia). The study also observed a downward trend in bone resorption risk and fracture risk.
3. New Exploration for Premenstrual Syndrome (PMS)
An open-label preliminary study in 2022 expanded the application of S-Equol to premenstrual symptoms. After two consecutive cycles of administration in 20 women with premenstrual symptoms, total symptom scores improved significantly, with a response rate (score reduction ≥50%) of 10.5%. The study also found a positive correlation between symptom improvement and Bifidobacterium in the gut microbiota, suggesting that gut microecology may play an important role.
III. Foundation of Safety: Favorable Tolerability and Pharmacokinetics
The efficacy of any intervention must be built on a foundation of safety.
1. Safety Verification at High Dosages
A pharmacokinetic study of S-Equol reported in 2011 tested single doses up to 320 mg and multiple doses up to 320 mg/day for 14 days in healthy volunteers. The conclusion clearly stated that all subjects tolerated S-Equol well, with no significant drug-related adverse events. It was rapidly absorbed, with exposure showing a linear relationship with dosage.
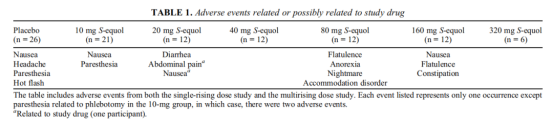
Sheet: Adverse events related or possibly related to the investigational product.
2. Preliminary Safety of Long-Term Use
In the aforementioned multiple studies lasting from 12 weeks to 12 months, no serious adverse events were reported, nor were abnormalities observed in blood biochemistry, cervical smears, mammograms, or other examinations.
No serious adverse events found in multiple 12-week to 12-month studies.
IV. Conclusion and Outlook
Synthesizing the above 10 clinical studies, a clear profile of S-Equol in menopausal care for women emerges:
-
Effective: S-Equol at 10 mg–20 mg/day demonstrates definitive clinical efficacy in relieving core menopausal symptoms such as hot flushes, night sweats, sleep disturbances, and musculoskeletal pain, especially for women unable to produce S-Equol endogenously.
-
Safe: Existing studies confirm its favorable safety and tolerability, with no serious adverse events observed even at dosages far exceeding the recommended dose, making it a promising mild alternative or complementary option to hormone replacement therapy (HRT).
-
Multifunctional: In addition to alleviating acute menopausal symptoms, S-Equol shows potential in maintaining bone health, improving cardiovascular parameters, and even anti-skin aging, meeting the long-term health management needs of menopausal women for both internal and external well-being.
Nevertheless, limitations remain in the sample size, study populations, and duration of existing research. Larger-scale studies with longer follow-up periods are needed to further clarify its long-term effects. Multiple large-scale randomized controlled trials are ongoing in Europe, the United States, and Asia, with more high-quality evidence-based data expected to be released in the next three years. Undeniably, as a natural molecule derived from soybeans, S-Equol, supported by robust evidence-based medical evidence, is providing a new, science-backed option for women suffering from menopausal discomfort.
About Our S-Equol
As a translational achievement of the above scientific evidence, our product boasts the following core advantages:
-
Advanced Technology
Adopting cutting-edge biosynthesis technology that mimics the conversion process of probiotics in the human gut, we achieve efficient, green, and large-scale production, with the entire process complying with food GMP requirements.
-
Superior Quality
The product is in the natural S-configuration, with purity and assay both exceeding 99%, presented as a white crystalline powder with consistent and stable quality.
-
Strict Quality Control
Stringent control of heavy metals, residual solvents, and other impurities, with microbial limits meeting high standards, ensuring safety and reliability.
We provide not only high-purity S-Equol raw materials but also a complete solution based on rigorous science. We sincerely invite partners in health products, functional foods, and innovative drugs to jointly develop this high-potential market and care for the health and quality of life of every woman through the power of science.




